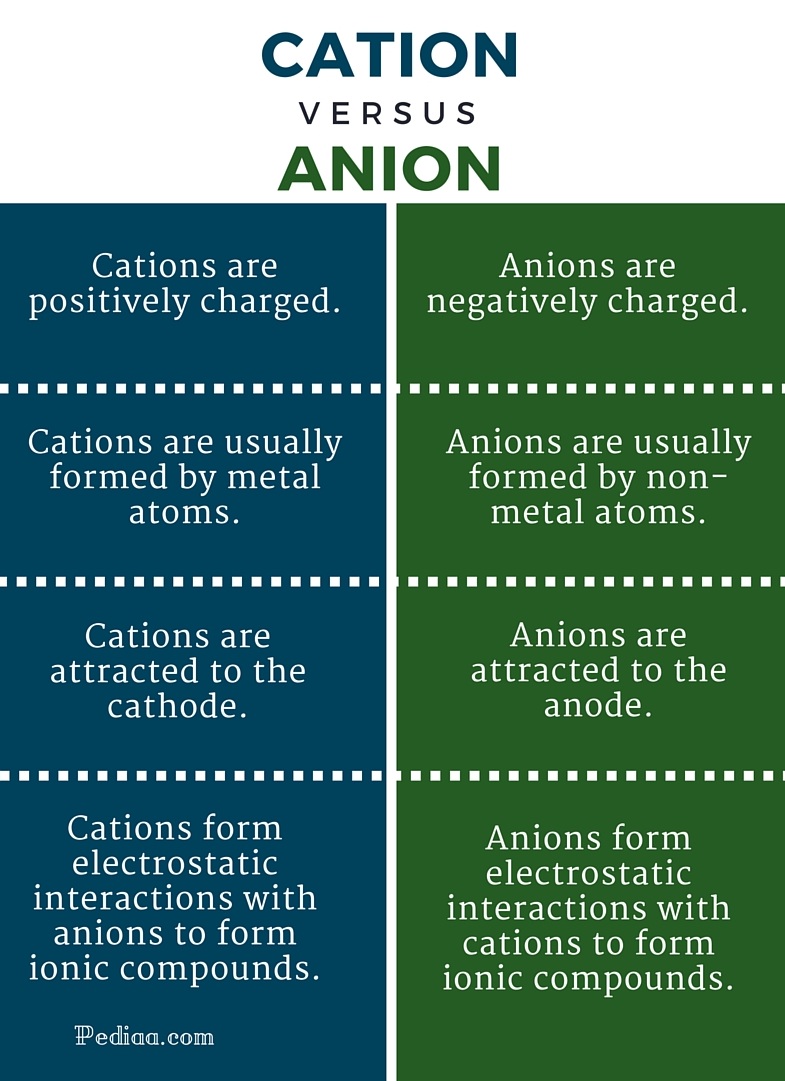
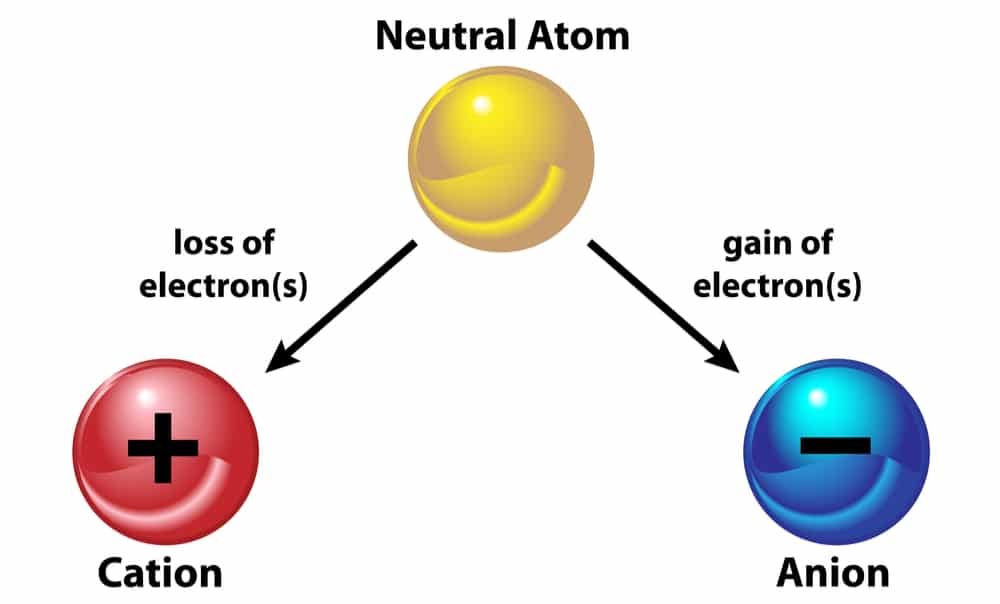
How Cations Are Formed - Positively charged ions formed as a result of losing electrons. A cation is formed from an atom that loses one or more electrons. Cations and anions form ionic bonds because of the. Cations are formed by the process of ionization when sufficient energy is given to the electron (by light of a high enough energy, for. Negatively charged ions formed as a result of gaining.
A cation is formed from an atom that loses one or more electrons. Cations and anions form ionic bonds because of the. Positively charged ions formed as a result of losing electrons. Cations are formed by the process of ionization when sufficient energy is given to the electron (by light of a high enough energy, for. Negatively charged ions formed as a result of gaining.
Negatively charged ions formed as a result of gaining. A cation is formed from an atom that loses one or more electrons. Positively charged ions formed as a result of losing electrons. Cations and anions form ionic bonds because of the. Cations are formed by the process of ionization when sufficient energy is given to the electron (by light of a high enough energy, for.
Diferencia entre aniónes y catiónes (con nomenclatura y ejemplos
A cation is formed from an atom that loses one or more electrons. Positively charged ions formed as a result of losing electrons. Cations and anions form ionic bonds because of the. Negatively charged ions formed as a result of gaining. Cations are formed by the process of ionization when sufficient energy is given to the electron (by light of.
Cations and Anions Difference between Cations and Anions
A cation is formed from an atom that loses one or more electrons. Negatively charged ions formed as a result of gaining. Positively charged ions formed as a result of losing electrons. Cations and anions form ionic bonds because of the. Cations are formed by the process of ionization when sufficient energy is given to the electron (by light of.
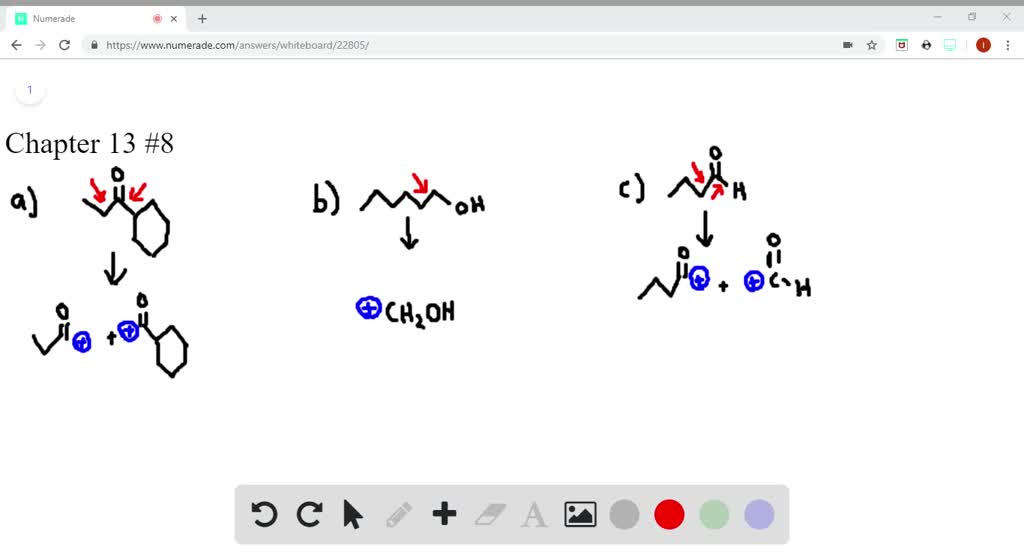
SOLVEDWhat cations are formed in the mass spectrometer by αcleavage of
A cation is formed from an atom that loses one or more electrons. Positively charged ions formed as a result of losing electrons. Negatively charged ions formed as a result of gaining. Cations and anions form ionic bonds because of the. Cations are formed by the process of ionization when sufficient energy is given to the electron (by light of.
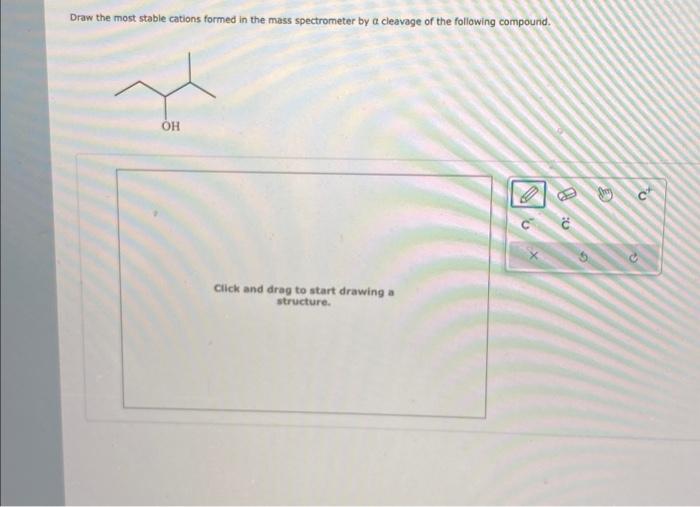
Solved Draw the most stable cations formed in the mass
A cation is formed from an atom that loses one or more electrons. Negatively charged ions formed as a result of gaining. Positively charged ions formed as a result of losing electrons. Cations and anions form ionic bonds because of the. Cations are formed by the process of ionization when sufficient energy is given to the electron (by light of.
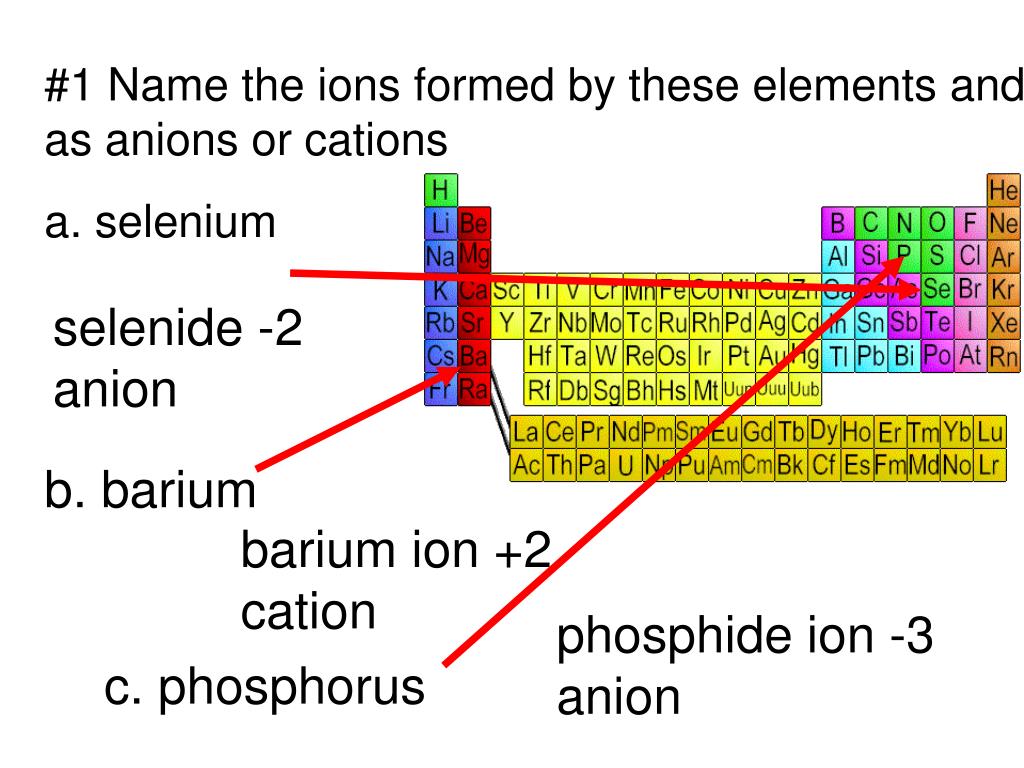
PPT 1 Name the ions formed by these elements and classify them as
Cations are formed by the process of ionization when sufficient energy is given to the electron (by light of a high enough energy, for. A cation is formed from an atom that loses one or more electrons. Negatively charged ions formed as a result of gaining. Cations and anions form ionic bonds because of the. Positively charged ions formed as.
Cations and Anions Definitions, Examples, and Differences
Cations are formed by the process of ionization when sufficient energy is given to the electron (by light of a high enough energy, for. A cation is formed from an atom that loses one or more electrons. Negatively charged ions formed as a result of gaining. Cations and anions form ionic bonds because of the. Positively charged ions formed as.
Solved Draw the most stable cations formed in the mass
Cations and anions form ionic bonds because of the. Cations are formed by the process of ionization when sufficient energy is given to the electron (by light of a high enough energy, for. Positively charged ions formed as a result of losing electrons. Negatively charged ions formed as a result of gaining. A cation is formed from an atom that.
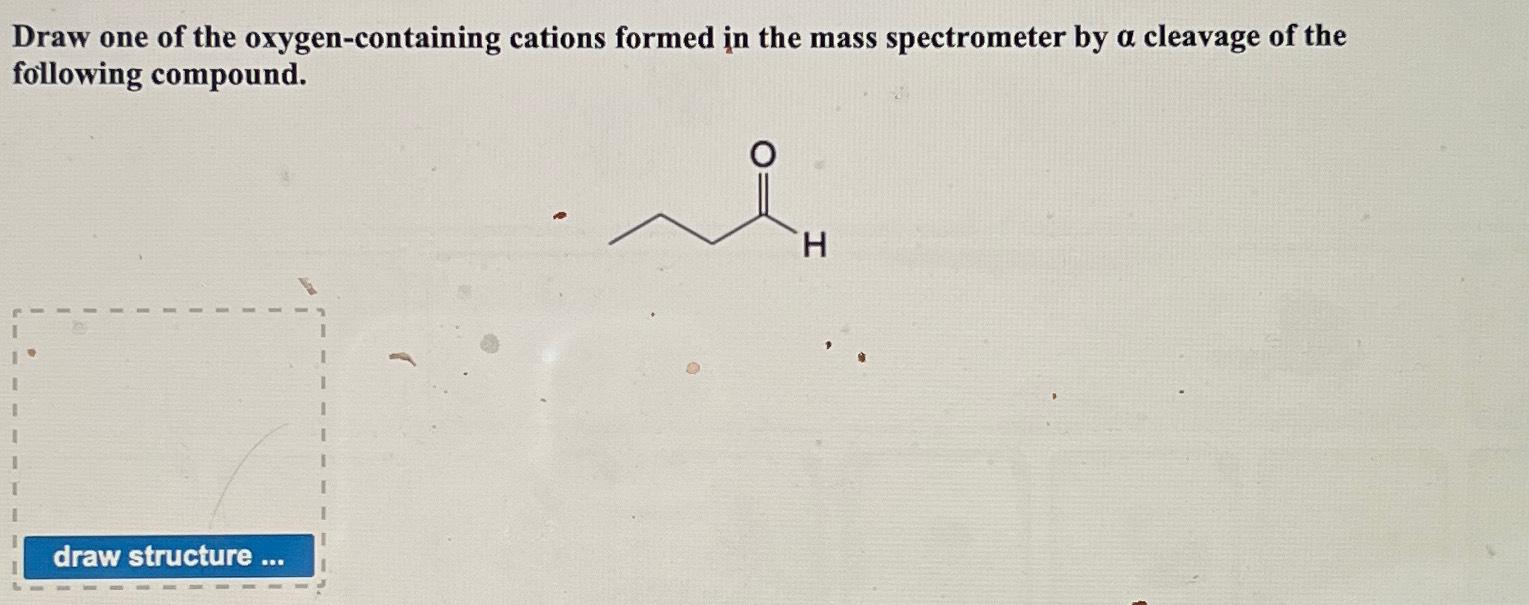
Solved Draw one of the oxygencontaining cations formed in
Cations and anions form ionic bonds because of the. Cations are formed by the process of ionization when sufficient energy is given to the electron (by light of a high enough energy, for. Positively charged ions formed as a result of losing electrons. A cation is formed from an atom that loses one or more electrons. Negatively charged ions formed.
Difference Between Cation and Anion
Cations are formed by the process of ionization when sufficient energy is given to the electron (by light of a high enough energy, for. A cation is formed from an atom that loses one or more electrons. Negatively charged ions formed as a result of gaining. Cations and anions form ionic bonds because of the. Positively charged ions formed as.
Amazing Tricks To Understanding Cations and Anions Key Concepts and
Cations and anions form ionic bonds because of the. Positively charged ions formed as a result of losing electrons. Cations are formed by the process of ionization when sufficient energy is given to the electron (by light of a high enough energy, for. Negatively charged ions formed as a result of gaining. A cation is formed from an atom that.
Cations And Anions Form Ionic Bonds Because Of The.
Positively charged ions formed as a result of losing electrons. Negatively charged ions formed as a result of gaining. Cations are formed by the process of ionization when sufficient energy is given to the electron (by light of a high enough energy, for. A cation is formed from an atom that loses one or more electrons.