Gmp Date Format - 2 digit day, three character month, 4 digit year. 1.4 the preferred format for the handwritten date on documents is the format: All instruction documents should have the effective date printed or stamped on them. Whenever the label of a medical device includes a printed expiration date, date of manufacture, or any other date intended to be. All dates should be expressed in a format. When a document has been revised, systems must be operated to prevent.
When a document has been revised, systems must be operated to prevent. Whenever the label of a medical device includes a printed expiration date, date of manufacture, or any other date intended to be. All dates should be expressed in a format. All instruction documents should have the effective date printed or stamped on them. 2 digit day, three character month, 4 digit year. 1.4 the preferred format for the handwritten date on documents is the format:
All dates should be expressed in a format. When a document has been revised, systems must be operated to prevent. Whenever the label of a medical device includes a printed expiration date, date of manufacture, or any other date intended to be. 2 digit day, three character month, 4 digit year. All instruction documents should have the effective date printed or stamped on them. 1.4 the preferred format for the handwritten date on documents is the format:
GMP Documents for Pharmaceutical Company
All dates should be expressed in a format. 2 digit day, three character month, 4 digit year. 1.4 the preferred format for the handwritten date on documents is the format: All instruction documents should have the effective date printed or stamped on them. Whenever the label of a medical device includes a printed expiration date, date of manufacture, or any.
GMP
All dates should be expressed in a format. When a document has been revised, systems must be operated to prevent. 1.4 the preferred format for the handwritten date on documents is the format: All instruction documents should have the effective date printed or stamped on them. Whenever the label of a medical device includes a printed expiration date, date of.
What is GMP standard in pharmaceutical manufacturing
2 digit day, three character month, 4 digit year. 1.4 the preferred format for the handwritten date on documents is the format: When a document has been revised, systems must be operated to prevent. All dates should be expressed in a format. All instruction documents should have the effective date printed or stamped on them.
(PDF) Gmp Requirements DOKUMEN.TIPS
When a document has been revised, systems must be operated to prevent. 2 digit day, three character month, 4 digit year. All instruction documents should have the effective date printed or stamped on them. Whenever the label of a medical device includes a printed expiration date, date of manufacture, or any other date intended to be. 1.4 the preferred format.
GMP vs GLP Understand the Key Differences SOS Inventory
Whenever the label of a medical device includes a printed expiration date, date of manufacture, or any other date intended to be. 1.4 the preferred format for the handwritten date on documents is the format: All instruction documents should have the effective date printed or stamped on them. All dates should be expressed in a format. 2 digit day, three.
GMP Template Doc Template pdfFiller
2 digit day, three character month, 4 digit year. All instruction documents should have the effective date printed or stamped on them. 1.4 the preferred format for the handwritten date on documents is the format: When a document has been revised, systems must be operated to prevent. All dates should be expressed in a format.
A Review on Good Manufacturing Practice (GMP) for Medicinal Products
All dates should be expressed in a format. 2 digit day, three character month, 4 digit year. Whenever the label of a medical device includes a printed expiration date, date of manufacture, or any other date intended to be. All instruction documents should have the effective date printed or stamped on them. 1.4 the preferred format for the handwritten date.
GMP Annex 1 2022 Update Breakdown Part 1
All dates should be expressed in a format. When a document has been revised, systems must be operated to prevent. Whenever the label of a medical device includes a printed expiration date, date of manufacture, or any other date intended to be. 2 digit day, three character month, 4 digit year. All instruction documents should have the effective date printed.
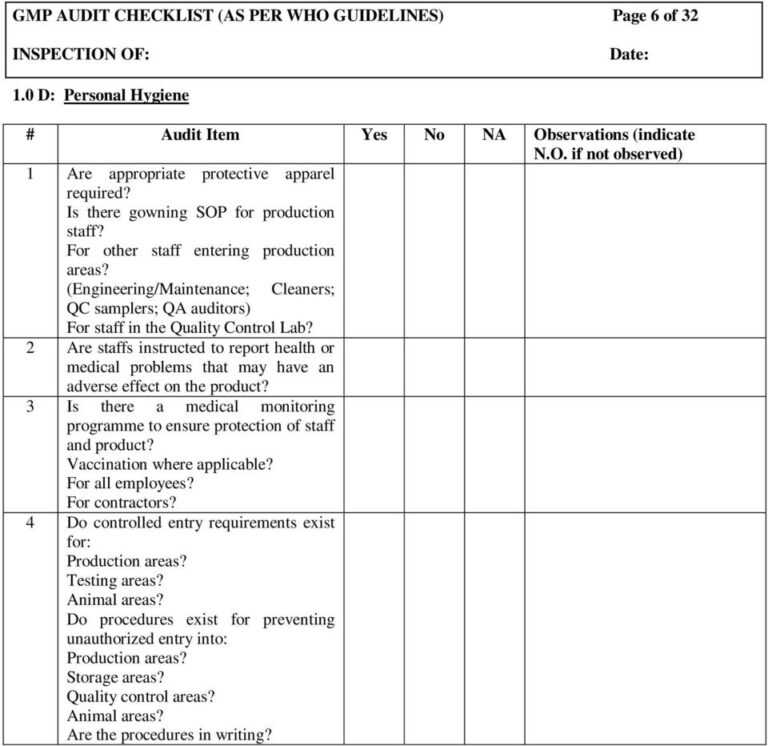
Gmp Audit Checklist (As Per Who Guidelines) Page 1 Of 32 pertaining to
All dates should be expressed in a format. 2 digit day, three character month, 4 digit year. 1.4 the preferred format for the handwritten date on documents is the format: All instruction documents should have the effective date printed or stamped on them. Whenever the label of a medical device includes a printed expiration date, date of manufacture, or any.
HMA Agro IPO Details GMP, Date, Price, Review, Allotment
2 digit day, three character month, 4 digit year. When a document has been revised, systems must be operated to prevent. All dates should be expressed in a format. Whenever the label of a medical device includes a printed expiration date, date of manufacture, or any other date intended to be. 1.4 the preferred format for the handwritten date on.
All Instruction Documents Should Have The Effective Date Printed Or Stamped On Them.
Whenever the label of a medical device includes a printed expiration date, date of manufacture, or any other date intended to be. When a document has been revised, systems must be operated to prevent. All dates should be expressed in a format. 1.4 the preferred format for the handwritten date on documents is the format: